Introduction: A Decision That Goes Beyond Price
Procurement decisions in healthcare are not just about issuing purchase orders. In reality, they directly affect patient outcomes, equipment uptime, maintenance costs, and even the reputation of the institution.
In today’s scenario, the discussion around Indian vs imported medical devices has become increasingly relevant. Hospitals must now balance cost efficiency with clinical performance and long-term reliability.
So, whether you are a procurement head, biomedical engineer, or clinic owner, one critical question arises:
Should you choose an Indian-made device or an imported one?
Over the past decade, India’s medical device ecosystem has matured significantly. This evolution is explained in detail in our guide on India’s medical device industry growth and trends.
Currently, many Indian manufacturers offer products approved by the Central Drugs Standard Control Organization (CDSCO), along with ISO 13485, CE, and US FDA certifications—making them globally competitive.
At the same time, imported devices—especially from Germany, Japan, the United States, and South Korea—continue to deliver advanced technology and strong clinical trust.
Therefore, this guide compares both options across cost, quality, compliance, service, and long-term value to support better procurement decisions.
1. The State of Indian Medical Device Manufacturing
India is currently the fourth-largest medical device market in Asia and is expected to reach USD 50 billion by 2030, as highlighted by the India Brand Equity Foundation (IBEF).
Moreover, regulatory reforms under the Medical Devices Rules, 2017 have strengthened quality standards. These regulations, governed by CDSCO, ensure proper classification, registration, and compliance.
In addition, initiatives such as the Production Linked Incentive (PLI) scheme have accelerated domestic manufacturing in areas like imaging systems, implants, diagnostics, and point-of-care technologies.
Consequently, manufacturing hubs in Pune, Bengaluru, Gujarat, and Himachal Pradesh are expanding rapidly.
However, it is important to note that not all manufacturers operate at the same level of maturity. Hence, careful evaluation remains essential.

2. COST & TOTAL COST OF OWNERSHIP (TCO)
At first glance, Indian medical devices appear significantly more affordable—typically costing 30–70% less upfront.
However, procurement decisions should not be based solely on purchase price. Instead, hospitals must evaluate the Total Cost of Ownership (TCO) over five years.
Specifically, this includes installation, training, consumables, AMC costs, spare parts, and downtime impact.
On the other hand, imported devices often involve proprietary consumables and foreign-currency-linked service contracts.
Typically, Indian manufacturers offer faster service response, local spare availability, and lower AMC costs.
3. Quality & Clinical Performance
In general, quality is the most critical factor in medical device procurement.
For Class A and B devices, many Indian products now match imported performance when properly certified, as seen in several categories of medical devices manufactured in India.
However, for more complex Class C and D devices, imported systems still maintain an advantage due to extensive R&D and long-term clinical data.
That said, this gap is steadily narrowing.
To ensure quality, hospitals should evaluate:
- Regulatory approvals
- Certification standards
- Clinical validation
- Sterilization compliance
4. Regulatory Compliance
To begin with, all medical devices sold in India must comply with Central Drugs Standard Control Organization (CDSCO) regulations.
However, compliance risks often arise due to unregistered suppliers, grey-market imports, or incomplete documentation.
Therefore, hospitals must treat regulatory compliance as a non-negotiable requirement rather than just a checklist item.
5. After‑Sales Service: The Operational Reality
Although procurement teams often focus on price, service reliability becomes crucial during daily operations.
For instance, delays in servicing ICU or critical care equipment can directly disrupt hospital workflows.
In most cases, Indian manufacturers offer faster service response, local spare availability, and lower AMC costs.
Before finalizing a purchase, hospitals should ask:
- What does the AMC include?
- What is the response time?
- Are engineers properly trained?
- Are spare parts locally available?
6. Lead Time & Supply Chain Reliability
Typically, imported devices require 8–20 weeks for delivery.
In contrast, domestic devices are usually delivered within 4–8 weeks.
For example, during the COVID-19 pandemic, global supply disruptions highlighted the importance of supply chain reliability.
As a result, hospitals now prioritize vendors who can ensure consistent and timely delivery.
7. Customisation & Local Adaptability
Another major advantage of Indian manufacturers is their ability to adapt to local conditions.
For example, many devices are designed to handle voltage fluctuations, environmental challenges, and regional training requirements.
Additionally, this flexibility is particularly beneficial for Tier-2 and Tier-3 healthcare facilities.
8. Quick Comparison Summary

Factor | Indian Devices | Imported Devices |
| Purchase Price | Lower upfront | Higher upfront |
| Total Cost of Ownership | Generally lower | May escalate |
| Regulatory Compliance | Mandatory CDSCO | Mandatory CDSCO |
| Clinical Performance | Strong in A/B devices | Strong in complex devices |
| Service Support | Faster local response | Brand dependent |
| Lead Time | Shorter | Longer |
| Customisation | Higher flexibility | Standardised design |
9. Category‑Based Practical Guidance
Overall, Indian devices perform well in:
- Surgical instruments
- Patient monitoring systems
- Digital X-ray
- Consumables
Imported devices dominate in:
- MRI systems
- Robotic surgery
- Advanced imaging
- Complex implants
👉 Therefore, always evaluate devices based on application—not origin.
10. Decision Framework for Hospitals
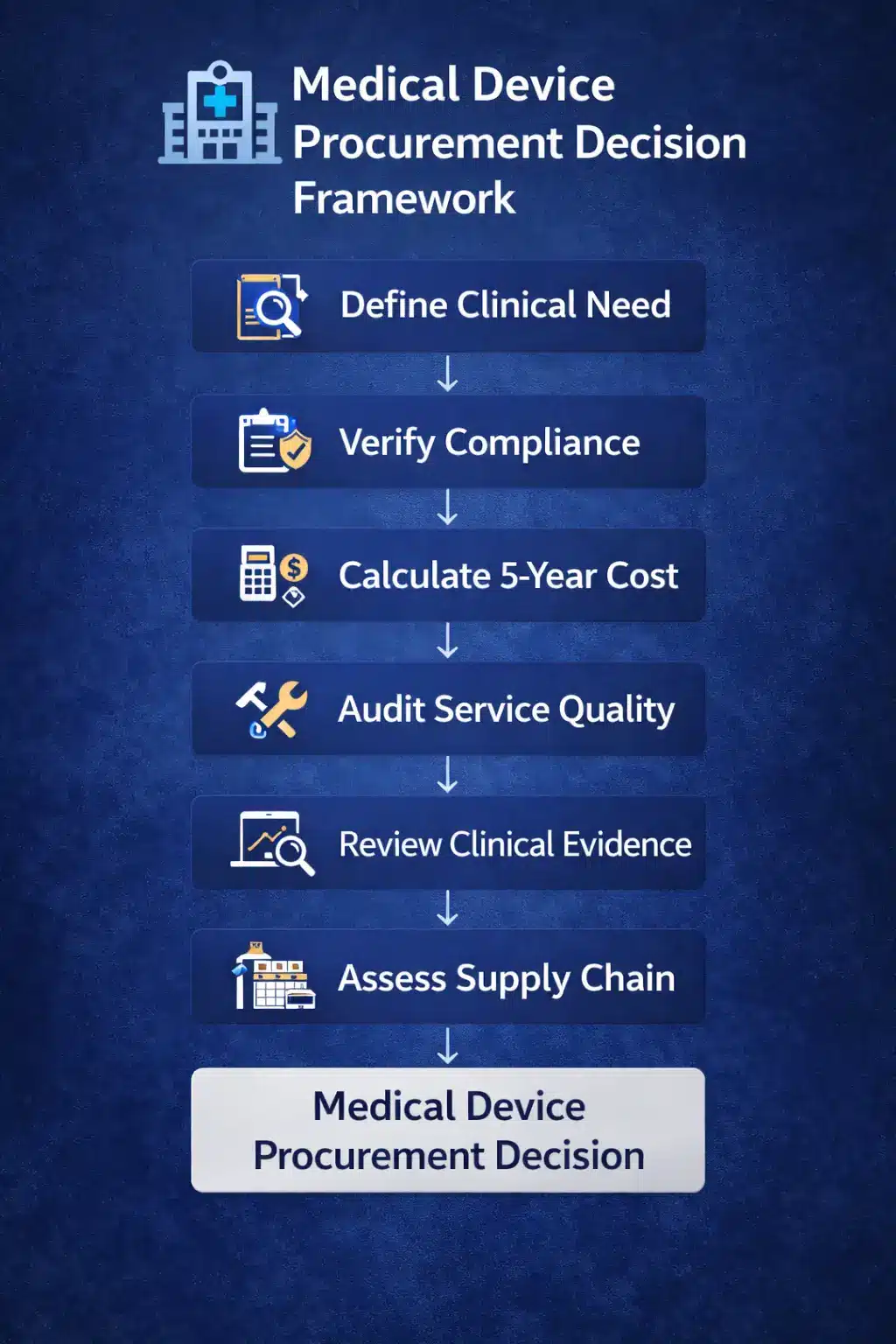
To ensure effective procurement, therefore, hospitals should follow a structured approach:
- Define clinical requirements
- Verify compliance
- Evaluate lifecycle cost
- Assess service support
- Review certifications
In practice, biomedical engineers play a critical role in this evaluation process. You can explore this further in our article on the role of biomedical engineers in hospitals.
11. Policy Environment & Strategic Direction
India’s medical device sector is supported by strong government initiatives.
For instance, programs like the PLI scheme and Make in India policy encourage domestic manufacturing, supported by initiatives such as Make in India.
However, these policies do not mandate procurement decisions, but they clearly indicate a long-term shift toward a stronger local ecosystem.
12. Common Myths — Clarified
Myth: Indian devices are inferior
Reality: Quality depends on certification and manufacturer capability
Myth: Imported devices offer better service
Reality: Service depends on local infrastructure
Myth: Doctors resist Indian devices
Reality: Proper validation and training improve adoption
Key Procurement Takeaways for Hospitals
- Firstly, Indian medical devices offer significant cost advantages in many product categories.
- However, imported equipment may still dominate in highly complex technologies such as advanced imaging and robotic surgery.
- In addition, after-sales service and spare part availability are critical for long-term operational reliability.
- Therefore, hospitals should evaluate five-year lifecycle cost rather than only the purchase price.
- Finally, regulatory compliance (CDSCO registration, ISO certification, and clinical validation) must always be verified.
Conclusion: Evidence Over Assumption
Ultimately, the choice between Indian and imported medical devices is not binary.
Indian manufacturers provide strong value across many categories.
However, imported devices still lead in highly complex technologies.
As a result, procurement decisions should be based on:
- Clinical performance
- Lifecycle cost
- Regulatory compliance
- Long-term reliability
In conclusion, the guiding principle is simple:
Decisions must be driven by evidence—not assumptions about country of origin.

