Where Engineering Precision Meets Patient
Introduction
Walk into any modern hospital and you will encounter an extraordinary range of medical technologies. These trends align with broader global healthcare technology developments.
These include a wide variety of medical devices used in modern hospitals — ventilators supporting critically ill patients, imaging systems detecting disease at early stages, patient monitors tracking vital signs continuously, and surgical equipment enabling complex procedures.
Behind the scenes, ensuring that all this technology works safely and reliably is a group of professionals who often remain invisible to patients but are essential to healthcare delivery:
Biomedical engineers.
Biomedical engineers operate at the intersection of medicine, engineering, and healthcare technology management.
Their responsibilities extend far beyond equipment repair. They play critical roles in:
- Medical equipment planning
- Safety assurance
- Procurement support
- Technology integration
- Lifecycle management
As healthcare becomes increasingly technology‑driven, the role of biomedical engineers has become more important than ever.
Industry Snapshot

Large tertiary hospitals may operate 1,500–2,500 medical devices. These devices range across categories explained in our guide on types of medical devices manufactured in India across departments including:
- Intensive care units
- Operating theatres
- Radiology departments
- Diagnostic laboratories
- Emergency departments
Managing this complex infrastructure requires structured technical oversight, which biomedical engineering teams provide.
Who Are Biomedical Engineers?
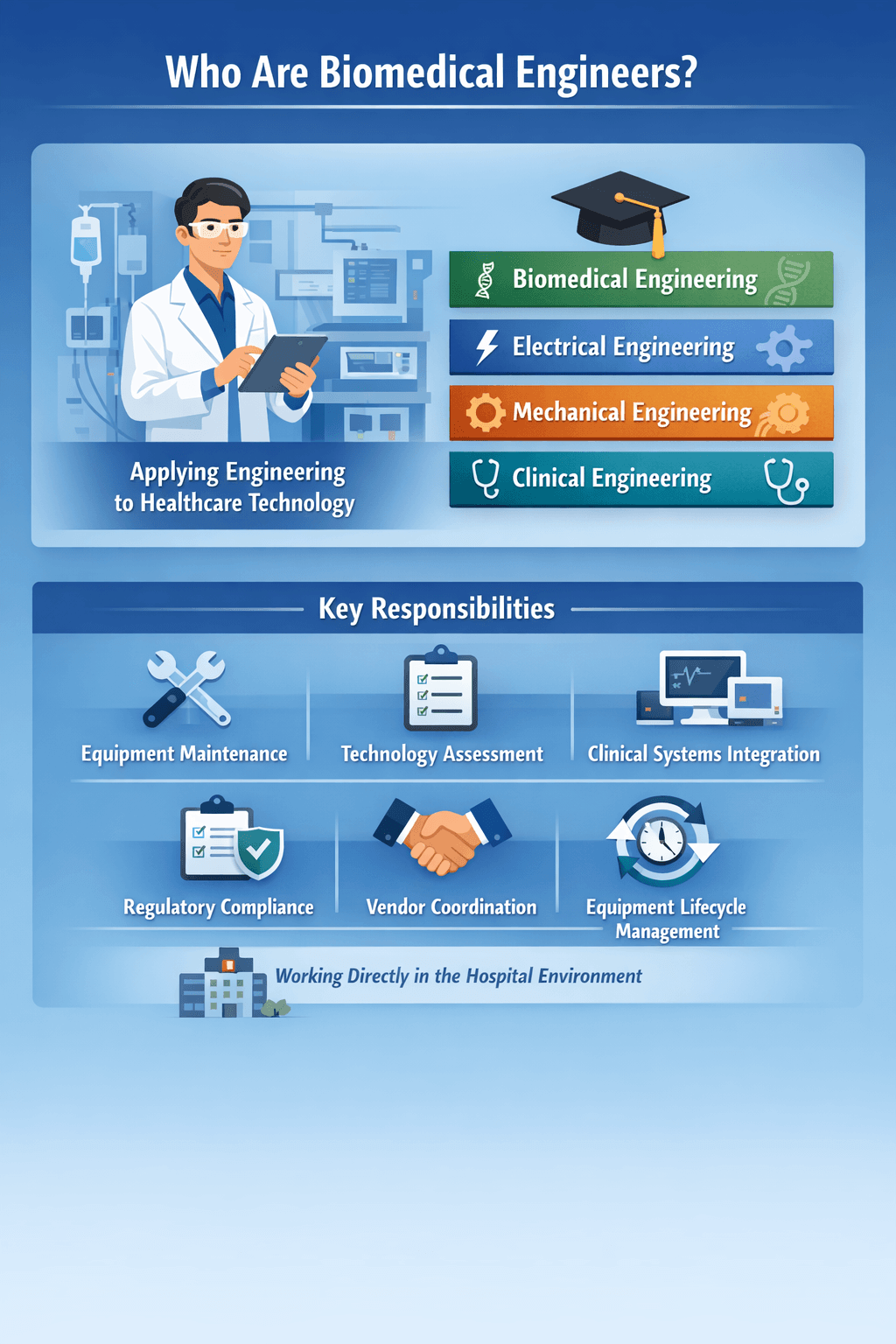
Biomedical engineers apply engineering principles to healthcare systems and medical technologies. The field is widely recognized under the biomedical engineering discipline overview.
In hospitals, professionals may hold degrees in:
- Biomedical engineering
- Electrical engineering
- Mechanical engineering
- Clinical engineering
Their responsibilities often include:
- Equipment maintenance and calibration
- Technology assessment and procurement support
- Clinical systems integration
- Regulatory compliance
- Vendor coordination
- Equipment lifecycle management
Unlike research biomedical engineers, hospital‑based biomedical engineers work directly within clinical environments.
Managing the Medical Equipment Lifecycle
This lifecycle management is influenced by the broader medical device industry in India.
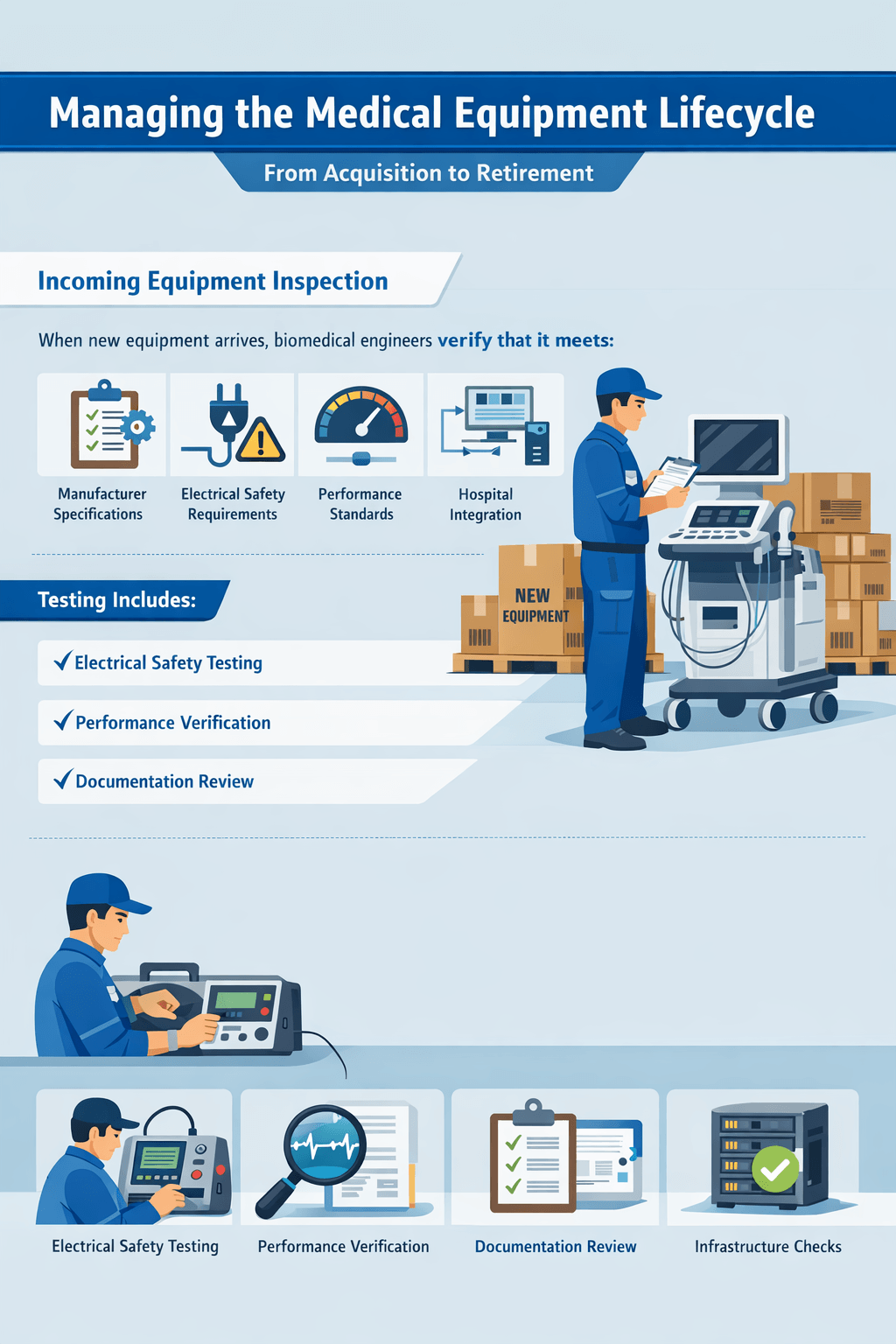
Biomedical engineers oversee the entire lifecycle of medical equipment, from acquisition to retirement.
Incoming Equipment Inspection
When new equipment arrives, biomedical engineers verify that it meets:
- Manufacturer specifications
- Electrical safety requirements
- Performance standards
- Hospital integration requirements
Testing may include:
- Electrical safety testing
- Performance verification
- Documentation review
- Infrastructure compatibility checks
Preventive Maintenance
Preventive maintenance programs ensure equipment remains reliable. These activities are part of broader hospital compliance and quality assurance systems. These practices follow medical equipment maintenance standards.
These programs include:
- Regular calibration
- Scheduled servicing
- Electrical safety testing
- Performance verification
Preventive maintenance helps avoid failures before they impact patient care.
Corrective Maintenance
When devices malfunction, biomedical engineers diagnose and coordinate repairs.
Rapid response is essential, particularly for critical care equipment used in ICU environments.
End‑of‑Life Management
As equipment ages, biomedical engineers evaluate whether devices should be repaired or replaced.
Decisions consider:
- Safety risks
- Maintenance costs
- Spare part availability
- Technology obsolescence
Ensuring Patient Safety and Regulatory Compliance
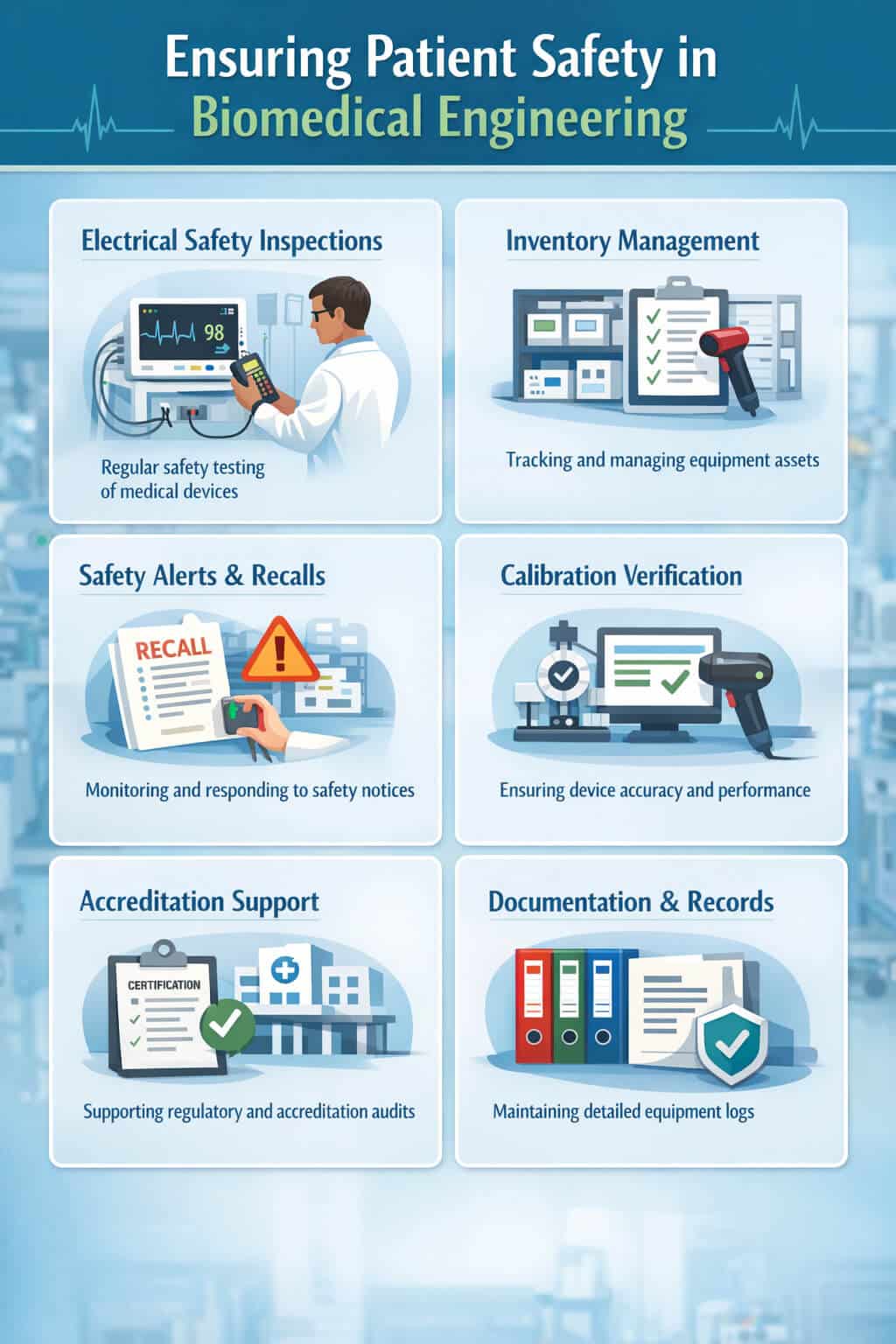
Patient safety lies at the core of biomedical engineering practice. This includes ensuring legal compliance and calibration of medical equipment in hospitals. These practices align with WHO patient safety guidelines.
Typical responsibilities include:
- Electrical safety inspections
- Equipment inventory management
- Monitoring safety alerts and recalls
- Calibration verification
- Supporting hospital accreditation processes
Proper documentation and equipment management systems help hospitals maintain compliance with regulatory and accreditation requirements. In India, this is governed by the Medical Device Rules 2017 (CDSCO).
Supporting Clinical Teams

Clinicians rely heavily on medical technology but may not always have detailed technical knowledge of device operation.
Biomedical engineers act as the technical bridge between clinicians and technology.
Their work includes:
- Training healthcare staff on equipment usage
- Troubleshooting during clinical procedures
- Investigating equipment‑related incidents
- Supporting advanced medical procedures
This collaboration allows clinicians to focus on patient care while engineers manage equipment safety and reliability.
Cybersecurity and Connected Medical Devices

Modern hospitals increasingly use connected medical devices as part of the Internet of Medical Things (IoMT). Many of these are part of the evolving ecosystem of modern medical devices in India. Global best practices are outlined in FDA medical device cybersecurity guidance
Devices such as:
- infusion pumps
- imaging systems
- patient monitoring systems
may be connected to hospital networks.
Biomedical engineers collaborate with IT teams to:
- assess cybersecurity risks during procurement
- ensure firmware and security updates are applied
- implement network protection measures
- respond to potential device security incidents
Protecting connected devices is essential for both patient safety and hospital data security.
Technology Integration and Innovation

Biomedical engineers also support hospitals in evaluating emerging technologies such as:
- Artificial intelligence‑assisted diagnostics
- Wearable patient monitoring devices
- Robotic surgical systems
- Remote patient monitoring platforms
- Point‑of‑care diagnostic technologies
Their role includes assessing whether new technologies can be safely integrated into existing clinical workflows.
Capital Planning and Procurement Support

Hospitals invest heavily in medical equipment each year.
Biomedical engineers support procurement decisions by:
- Evaluating technical specifications
- Comparing manufacturer options
- Assessing service requirements
- Identifying infrastructure needs
- Estimating lifecycle costs
Their analysis ensures procurement decisions align with clinical, technical, and financial considerations. These decisions are supported by health technology assessment frameworks. Engineers play a key role in Indian vs imported medical device procurement decisions.
Challenges Biomedical Engineers Face
Despite their importance, biomedical engineers often face several challenges:
- Managing large device inventories with limited staff
- Budget constraints for equipment upgrades
- Rapid technological change
- Increasing regulatory expectations
- Balancing urgent clinical needs with technical processes
Addressing these challenges is essential for strengthening healthcare technology management systems.
Conclusion
Biomedical engineers may not always be visible in patient care environments, but their work directly supports the safety and effectiveness of modern healthcare.
From managing equipment lifecycles and ensuring regulatory compliance to supporting clinicians and integrating new technologies, biomedical engineers play a vital role in hospital operations.
As healthcare technology continues to evolve, the contribution of biomedical engineers will remain essential in ensuring that medical devices function safely, efficiently, and in the best interest of patient care.
Related Reading:

