A Practical Guide for Hospitals & Medical Device Management (India Focus)
🔷 Introduction: Why This Matters More Than Ever
In today’s healthcare ecosystem, legal compliance in healthcare India has become a critical priority. Hospitals must implement strong hospital compliance systems where every decision impacts patient safety.
From a simple thermometer to a complex ventilator, every medical device must be:
- Legally compliant
- Quality assured
- Properly calibrated
These devices are part of the broader ecosystem of medical devices manufactured in India.
Yet, many hospitals still treat these as checklist activities, not strategic priorities.
👉 That’s where problems begin.
⚖️ Understanding Legal Compliance in Healthcare
Legal compliance in healthcare ensures hospitals meet NABH compliance requirements and follow healthcare regulations in India. These regulations are closely linked with the growth of India’s medical device industry and policy framework.
Use entity references:
- CDSCO – Medical device regulation
- NABH – Hospital accreditation & quality standards
- BIS – Product standards
- AERB – Radiation equipment safety

🧾 Why Compliance is Non-Negotiable
- Prevents legal penalties and shutdown risks
- Ensures eligibility for insurance & government schemes
- Builds trust with patients and stakeholders
👉 Hospitals that ignore hospital compliance India standards risk penalties and safety issues.
🔍 Quality Assurance in Hospitals: Ensuring Medical Device Quality
Quality assurance in hospitals ensures that medical devices perform safely and consistently. This involves a series of structured validation processes across the device lifecycle. These processes are also managed as part of medical equipment lifecycle management in hospitals.
🧩 Key QA Processes in Hospitals
- Incoming Inspection – Checking device quality at delivery
- Installation Qualification (IQ) – Proper setup verification
- Operational Qualification (OQ) – Functional validation
- Performance Qualification (PQ) – Real-world performance check

These are part of a structured hospital quality management system.
🎯 Why QA Matters
- Reduces equipment failure
- Improves clinical outcomes
- Extends device lifespan
- Strengthens NABH compliance
👉 QA is not about fixing problems—it’s about preventing them.
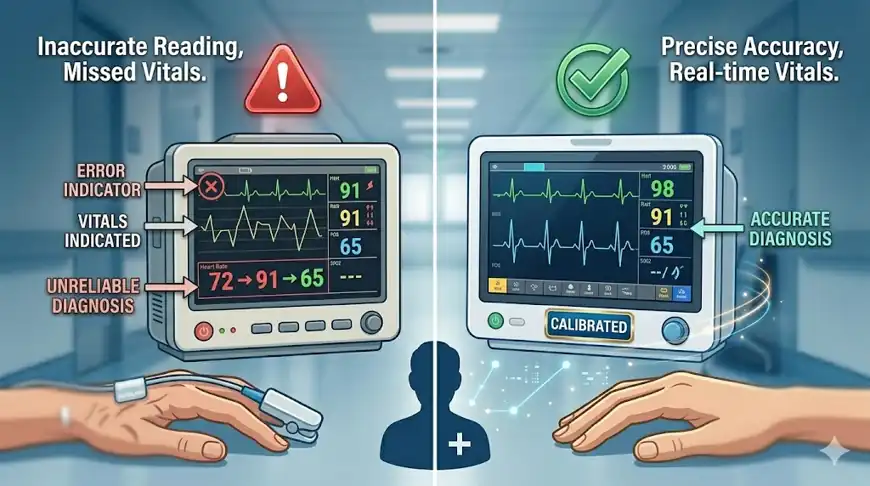
⚙️ Calibration of Medical Equipment: The Silent Lifesaver
Medical equipment calibration ensures accuracy in diagnosis and treatment.
📌 Why Calibration is Critical
- Even small errors can lead to wrong diagnosis or treatment
- Proper calibration of medical devices directly impacts patient safety.
- Mandatory for accreditation & audits
🏥 Common Equipment That Requires Calibration
- Patient monitors
- Infusion pumps
- Ventilators
- Defibrillators
- Laboratory analyzers
⏱️ Calibration Frequency
- Daily (some lab equipment)
- Monthly / Quarterly
- Annually
- After repair or relocation
👉 Without calibration, equipment accuracy in hospitals cannot be guaranteed. Biomedical engineers play a key role in ensuring this, as explained in the role of biomedical engineers in hospitals.
👨🔬 Role of Biomedical Engineering in Hospitals
Biomedical engineering in hospitals plays a crucial role in ensuring compliance and safety.
🔑 The role of biomedical engineers includes…
- Device selection during procurement
- Ensuring regulatory compliance
- Managing calibration schedules
- Preventive maintenance planning
- Supporting audits and inspections
👉They are essential for effective hospital equipment management. Their role becomes especially critical during medical device procurement decisions in hospitals.
🛒 Why Compliance Must Start at Procurement
Medical device procurement in India must focus on compliance from the beginning. Hospitals must evaluate options carefully using a comparison between Indian and imported medical devices.
Most hospitals make a critical mistake:
👉 They think compliance starts after purchase.
✔️A structured compliant procurement healthcare approach includes:
- Verify CDSCO / CE / FDA approvals
- Evaluate vendor service & calibration support
- Check spare parts availability
- Consider lifecycle cost (not just price)
👉Smart hospital equipment purchasing reduces compliance risks.
📊 Documentation: The Most Ignored Risk Area
Hospital documentation compliance ensures traceability and audit readiness.
In healthcare:
“If it’s not documented, it didn’t happen.”
📁 Must-Have Records
Maintaining accurate medical equipment records is essential.
- Equipment inventory
- Calibration certificates
- Maintenance logs
- Breakdown reports
- Vendor agreements
💡 Pro Tip:
Use a CMMS (Computerized Maintenance Management System) hospital system to digitize records. This becomes essential when managing a large inventory of medical devices used in modern hospitals.
⚠️ Common Challenges Hospitals Face
Hospitals often face multiple healthcare compliance challenges.
- Lack of trained biomedical staff
- Poor calibration tracking systems
- Budget constraints
- Over-dependence on vendors
- Incomplete documentation
👉These hospital compliance issues in India can impact patient safety.
✅ Best Practices for Hospitals (MIIMD Recommended)
Implementing healthcare compliance best practices ensures long-term success.
- Implement CMMS for equipment tracking
- Create annual calibration plans
- Partner with NABL-accredited labs
- Train staff regularly
- Align procurement with compliance
This leads to continuous hospital quality improvement.
🚀 Future Trends in Medical Device Management
The future of digital healthcare compliance is transforming hospitals.
AI in hospital equipment management enables predictive maintenance.- IoT-enabled smart devices
- Digital calibration records
- Automated compliance systems
These advancements are part of the evolving medical device industry in India.
👉 This shift is shaping smart hospitals in India.
🧠 Final Thought
Legal compliance, quality assurance, and calibration are not separate activities.
These pillars directly improve patient safety in hospitals.
Hospitals that integrate them into daily operations will achieve:
✔ Better patient outcomes
✔ Strong regulatory standing
✔ Higher operational efficiency
✔ Long-term cost savings
They also enhance overall healthcare operational efficiency.

