Introduction: A Decision That Goes Beyond Price
Every procurement decision in a healthcare facility goes far beyond issuing a purchase order. It affects patient outcomes, equipment uptime, maintenance budgets, and ultimately the reputation of the institution.
The debate around Indian vs imported medical devices has become increasingly relevant as hospitals focus on balancing cost, clinical performance, and long-term operational reliability.
Whether you are a procurement head at a multi‑specialty hospital, a clinic owner in a Tier‑2 city, or a biomedical engineer evaluating capital equipment, the key question often arises:
Should we choose an Indian‑made medical device or an imported one?
The answer is rarely black and white.
India’s medical device ecosystem has matured significantly over the past decade. The country’s MedTech sector is evolving from import dependence toward domestic manufacturing capability and global exports. (You can read a deeper industry analysis in our article on India’s Medical Device Industry: Growth, Opportunities & Challenges.). The domestic market is now estimated at USD 12–15 billion and growing at approximately 15% CAGR, with increasing manufacturing capability and policy support. According to the Indian medical device market report, the sector is growing rapidly.
Many domestic manufacturers now produce CDSCO‑approved, ISO 13485‑certified, CE‑marked, and even US FDA‑cleared products that compete globally.
At the same time, imported devices — particularly from Germany, Japan, the United States, and South Korea — bring decades of engineering refinement, global brand equity, and established clinical confidence.
This guide evaluates both options across the dimensions that truly matter in real‑world procurement:
- Total Cost of Ownership
- Clinical performance
- Regulatory compliance
- After‑sales service
- Supply chain reliability
- Long‑term institutional value
The goal is not to promote one side — but to support informed, defensible decision‑making.
1. The State of Indian Medical Device Manufacturing
India is currently the fourth-largest medical device market in Asia. A broader analysis of the sector’s evolution, policy drivers, and export growth can be explored in our article India’s Medical Device Industry: Growth, Opportunities & Challenges.with projections estimating growth to USD 50 billion by 2030.
Regulatory reforms under the Medical Devices Rules, 2017 have strengthened quality oversight and aligned Indian regulation with international device classification frameworks. These regulations are governed by the Central Drugs Standard Control Organization (CDSCO). CDSCO now mandates device classification, registration, and compliance aligned with international frameworks. Ensuring compliance with these regulations is a critical function of biomedical engineers in hospitals.
The Production Linked Incentive (PLI) Scheme, with an outlay exceeding ₹3,420 crore, has accelerated domestic investment in:
- Imaging systems
- Implantable devices
- In‑vitro diagnostics
- Point‑of‑care technologies
Manufacturing clusters in Pune, Bengaluru, Himachal Pradesh, Uttarakhand, and Gujarat continue expanding capacity.

Several Indian companies now export to regulated global markets — an indicator of growing compliance with international standards.
However, maturity across the ecosystem is not uniform, which makes careful evaluation essential.
Indian vs Imported Medical Devices: Key Procurement Differences for Hospitals
2. Head‑to‑Head Comparison: What Actually Matters
2.1 Cost & Total Cost of Ownership (TCO)
Indian medical devices often cost 30–70% less upfront compared to imported equivalents.
However, experienced procurement teams evaluate five‑year Total Cost of Ownership (These evaluations are often carried out through medical equipment lifecycle management practices), which includes:
- Installation and commissioning
- Staff training
- Consumables
- Annual maintenance contracts (AMC)
- Spare parts availability
- Equipment downtime impact
- Exchange rate exposure
Imported systems frequently involve:
- Proprietary consumables
- Foreign‑currency AMC pricing
- OEM‑restricted spare parts
Indian manufacturers often provide:
- Locally priced AMCs
- Faster spare availability
- Reduced currency risk
For high‑volume consumables — such as IV sets, syringes, surgical gloves, ECG electrodes — importing rarely makes economic sense when certified domestic alternatives exist.
2.2 Quality & Clinical Performance
Quality remains the most critical factor — and also the most nuanced.
For Class A and Class B devices (low‑to‑moderate risk categories such as disposables, instruments, and basic diagnostics), leading Indian manufacturers frequently match imported clinical performance when properly certified.
For Class C and Class D devices (complex imaging systems, robotic surgery platforms, advanced implantables), global manufacturers often maintain a technological advantage due to long‑term R&D investment and extensive clinical outcome data.
However, the gap is gradually narrowing.
A practical observation across many hospitals is that quality variation can exist among smaller domestic manufacturers, making due diligence essential.
Procurement teams should always verify:
- CDSCO registration
- ISO 13485 certification. These certifications align with global standards such as ISO 13485 medical device quality management.
- CE or US FDA clearance (where applicable)
- Clinical performance evidence
- Biocompatibility and sterilisation validation
- Shelf‑life documentation
Quality ultimately depends more on manufacturer discipline and regulatory compliance than country of origin.
2.3 Regulatory Compliance
All medical devices sold in India — domestic or imported — must comply with regulations issued by the Central Drugs Standard Control Organization, the national regulatory authority responsible for medical device oversight.
Compliance risks typically arise from:
- Unregistered small suppliers
- Grey‑market importers
- Incomplete regulatory documentation
Regulatory compliance should be treated as a non‑negotiable entry criterion, not a scoring parameter.
2.4 After‑Sales Service: The Operational Reality
Service reliability is often underestimated during procurement but becomes critical during operations. This highlights the importance of hospital equipment maintenance and engineering support.
In real hospital environments, even a 48‑hour delay in servicing ICU equipment can disrupt workflows and increase operational stress.
Indian manufacturers often offer:
- Faster regional engineer deployment
- Locally stocked spare parts
- Lower AMC costs
Imported brands vary widely:
- Premium OEMs often have strong service networks
- Mid‑tier imports may rely on distributors, creating service variability
Key service questions procurement teams should ask:
- What is the guaranteed service response time?
- Are engineers factory‑trained?
- Are spare parts stocked within India?
- What is the Mean Time Between Failures (MTBF)?
- What exactly does the AMC include?
In many cases, service quality outweighs brand perception in long‑term satisfaction.
2.5 Lead Time & Supply Chain Reliability
Imported capital equipment may require 8–20 weeks for delivery.
Domestic equipment may ship within 4–8 weeks.
The COVID‑19 pandemic demonstrated the vulnerability of import‑dependent supply chains. The pandemic highlighted global healthcare supply chain disruptions. Hospitals relying on overseas suppliers experienced delays in:
- PPE
- ventilators
- diagnostic equipment
Supply chain resilience is now considered a strategic procurement factor.
2.6 Customisation & Local Adaptability
Indian manufacturers often design products considering local conditions such as:
- Voltage fluctuations
- UPS compatibility
- Environmental factors
- Regional training requirements
This adaptability can be especially valuable in Tier‑2 and Tier‑3 healthcare facilities.
3. Quick Comparison Summary

Factor | Indian Devices | Imported Devices |
| Purchase Price | Lower upfront | Higher upfront |
| Total Cost of Ownership | Generally lower | May escalate |
| Regulatory Compliance | Mandatory CDSCO | Mandatory CDSCO |
| Clinical Performance | Strong in A/B devices | Strong in complex devices |
| Service Support | Faster local response | Brand dependent |
| Lead Time | Shorter | Longer |
| Customisation | Higher flexibility | Standardised design |
4. Category‑Based Practical Guidance
Strong Value from Indian Manufacturers
- Surgical instruments and disposables
- Patient monitoring systems
- Semi‑automated diagnostics
- Digital X‑ray for primary care
- High‑volume consumables
Segments Where Imports Still Lead
- High‑field MRI systems
- Robotic surgical platforms
- Advanced imaging ecosystems
- Complex implantable devices
Procurement decisions should always be device‑specific rather than origin‑specific.
5. A Structured Decision Framework
Hospitals should adopt a structured evaluation approach: Many hospitals follow health technology assessment methodologies.
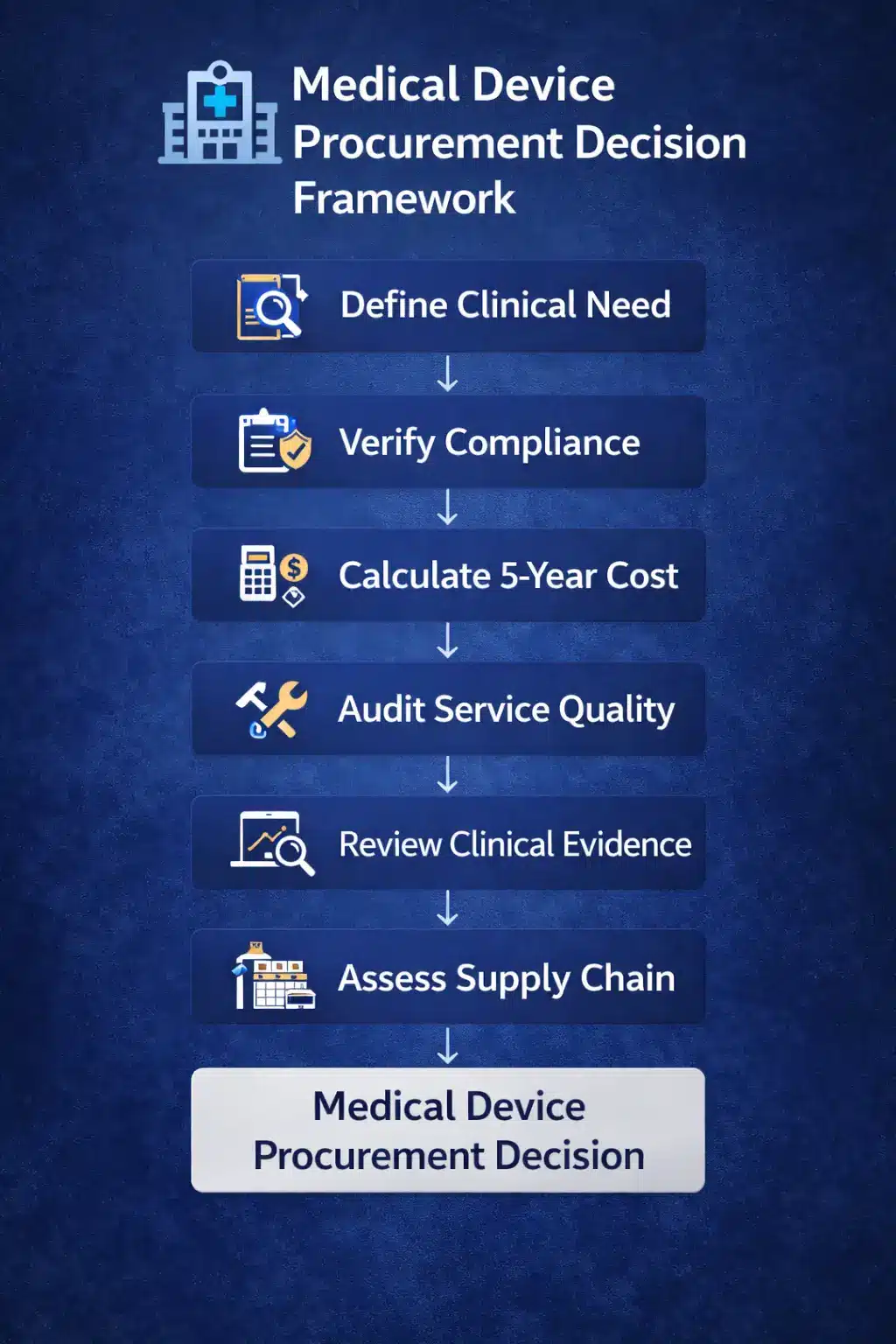
- Define the exact clinical use case
- Verify regulatory compliance
- Calculate five‑year total cost of ownership
- Audit service ecosystem and response times
- Review clinical evidence and certifications
- Assess supply chain reliability
- Apply transparent scoring criteria
Biomedical engineers play a critical role in this process, ensuring technical evaluation aligns with clinical and operational needs. Their responsibilities are detailed in our article on the role of biomedical engineers in hospitals.
6. Policy Environment & Strategic Direction
India’s medical device sector is also being shaped by evolving policy initiatives aimed at strengthening domestic manufacturing and reducing long-term import dependence.
Government programs such as the Production Linked Incentive Scheme for Medical Devices encourage investment in high-value device manufacturing segments including imaging systems, advanced diagnostics, and critical care equipment. The scheme provides financial incentives to manufacturers who expand domestic production capacity and achieve defined sales growth targets. Details are available under the PLI scheme for medical devices.
In parallel, policy frameworks such as Public Procurement (Make in India) preference policies and calibrated adjustments in Basic Customs Duty on selected medical device imports are designed to support the development of a stronger domestic ecosystem.
These initiatives do not mandate procurement choices for hospitals or healthcare institutions. However, they clearly signal a strategic national direction toward building a more resilient and self-reliant medical device industry while maintaining access to global innovation where required.they signal future ecosystem development.
7. Common Myths — Clarified
Myth: Indian devices are inherently inferior
Reality: Quality depends on certification, manufacturer capability, and compliance.
Myth: Imported devices always provide better service
Reality: Service quality often depends on distributor support.
Myth: Doctors resist Indian devices
Reality: Structured clinical evaluation increases acceptance.
Key Procurement Takeaways for Hospitals
- Indian medical devices offer significant cost advantages in many product categories.
- Imported equipment may still dominate in highly complex technologies such as advanced imaging and robotic surgery.
- After-sales service and spare part availability are critical factors for long-term operational reliability.
- Hospitals should evaluate five-year lifecycle cost rather than only the purchase price.
- Regulatory compliance (CDSCO registration, ISO certification, and clinical validation) must always be verified.
Conclusion: Evidence Over Assumption
The debate between Indian and imported medical devices is often framed as a binary choice. In reality, procurement should focus on:
Clinical performance
Lifecycle cost
Service reliability
Regulatory compliance
Institutional needs
In many segments today, Indian manufacturers represent a competitive and practical choice.
In highly complex capital equipment segments, imported solutions may remain appropriate.
The most important shift is this:
Procurement decisions must be guided by structured evaluation — not assumptions based on origin.
Ultimately, the choice between Indian vs imported medical devices should be based on evidence-driven evaluation rather than assumptions about origin.t assumptions based on origin.
Frequently Asked Questions (FAQ)
1. Are Indian medical devices reliable for hospitals?
Yes. Many Indian medical device manufacturers now produce CDSCO-approved, ISO 13485-certified, and internationally compliant products that are widely used in hospitals. For several device categories such as disposables, patient monitoring systems, and basic diagnostics, Indian devices can match the quality and performance of imported alternatives when sourced from reputable manufacturers.
2. Why do hospitals still import medical devices?
Hospitals often import devices for highly complex technologies such as high-field MRI systems, robotic surgical platforms, advanced imaging ecosystems, and certain implantable devices. Global manufacturers in these segments typically have decades of research and clinical data supporting their technologies.
3. What factors should hospitals consider when choosing between Indian and imported medical devices?
Hospitals should evaluate several factors including clinical performance, regulatory compliance, total cost of ownership, after-sales service support, spare part availability, and supply chain reliability. Procurement decisions should focus on long-term operational value rather than only the purchase price.
4. Are Indian medical devices more cost-effective than imported devices?
In many product categories, Indian medical devices offer significant cost advantages. Domestic manufacturers often provide lower upfront pricing, locally priced annual maintenance contracts, and faster spare part availability, which can reduce the overall lifecycle cost for hospitals.
5. Is the Indian medical device industry growing?
Yes. The Indian medical device market is expanding rapidly and is projected to reach approximately USD 50 billion by 2030. Government initiatives, increased domestic manufacturing capacity, and growing healthcare demand are driving this growth.
/Explore more:

